Microfluidic Cell Assay
Photolithography (a, b, c) and Soft Lithography (d, e, f):
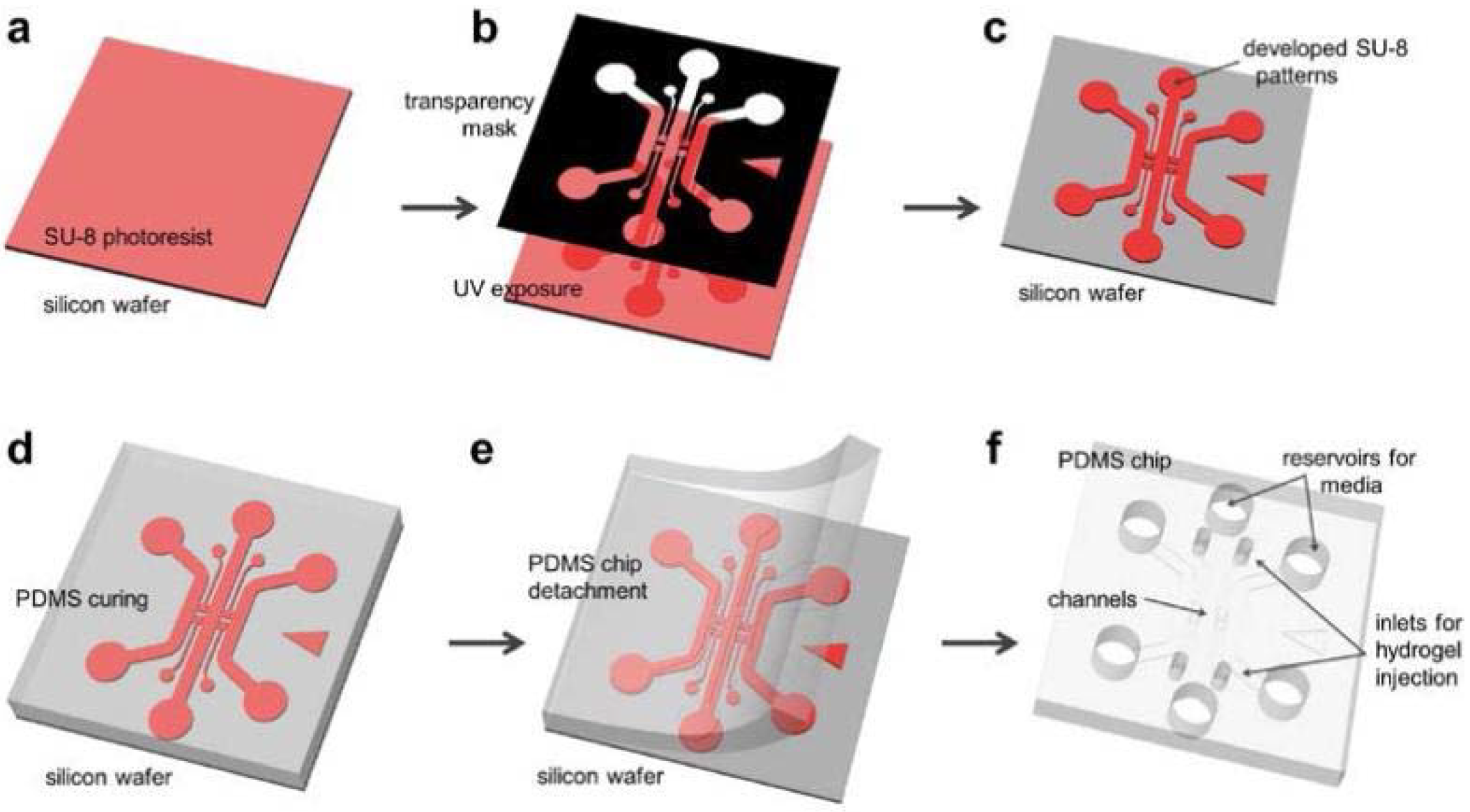
[1] SU-8 is spin-coated and pre-baked on a bare wafer;
[2] Use a transparency photo mask (black), ultraviolet (UV) light is exposed on the SU-8;
[3] Exposed SU-8 is then post-exposure baked and developed to define channel patterns;
[4] Polydimethylsiloxane (PDMS) mixed solution is poured on the wafer and cured;
[5] Cured PDMS is then peeled from the wafer;
[6] Device is trimmed, punched and autoclaved ready for assembly.
Hydrogel IncorporatingMicrofluidic Assay:
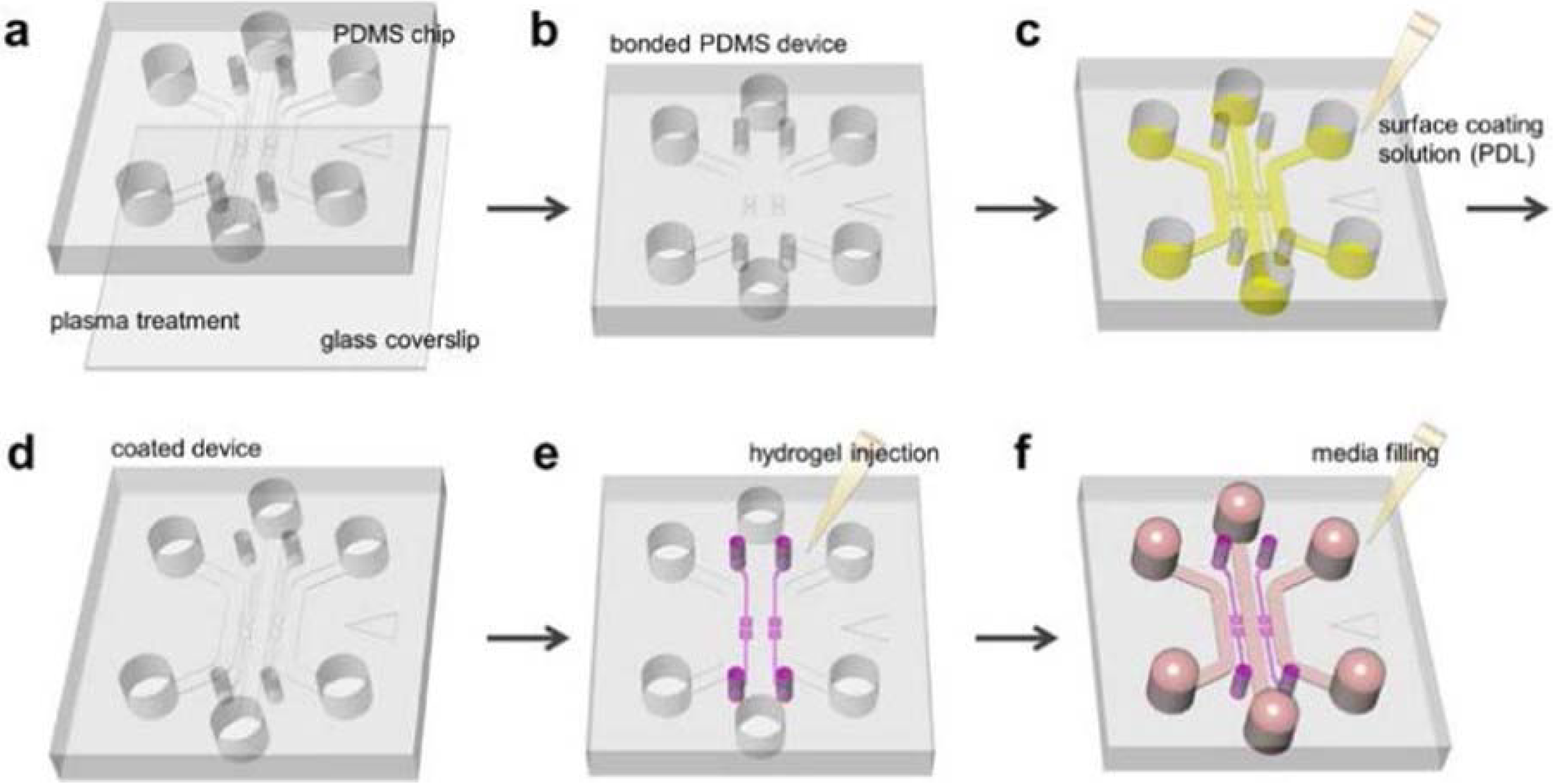
[1] Autoclaved PDMS device and coverslip are assembled with plasma treatment;
[2] To close the microfluidic channels;
[3] Surface coating solution (PDL) solution is filled and device is placed in an incubator;
[4] After washing and aspiration, coated device is stored in dry oven for 24 hours to render the microfluidic channel surface hydrophobic;
[5] Hydrogel is filled into the hydrogel region;
[6] Medium is added into the microfluidic channel.
Device is ready for cell seeding in incubator.
Cell Culturein Microfluidic Assay:
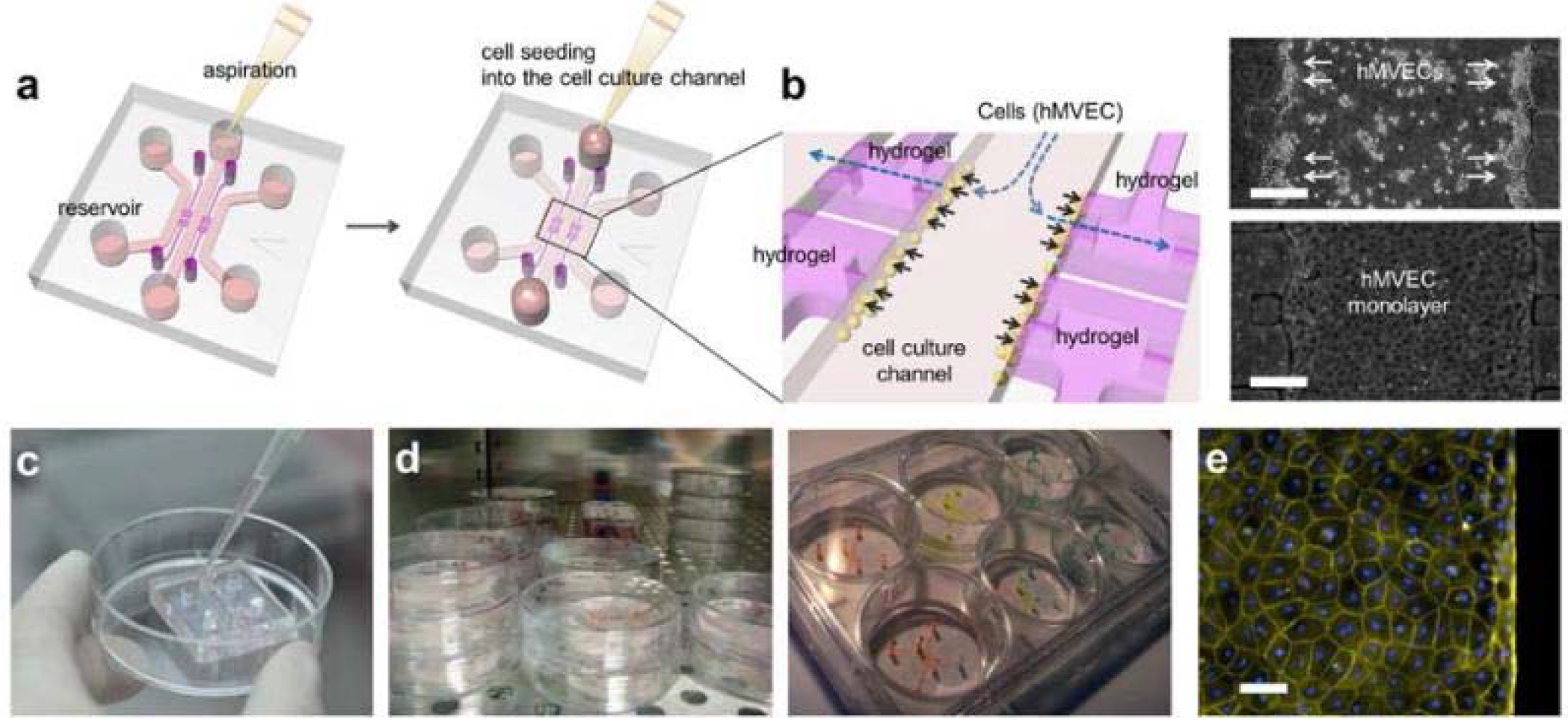
[1] After aspiration of medium in the reservoir, 50~60 μL of the human microvascular endothelial cell (hMVEC) suspension is added into one reservoir connected to the center channel.
[2] Hydrostatic pressure difference in the reservoirs pushes the suspended hMVECs onto the collagen gel to facilitate attachment. hMVECs right after seeding (right top) and tight monolayer at 1 day (right bottom). Scale bar indicates 250 microns.
[3] To add cell suspension or media into the reservoir, use a pipette and gently inject medium into one reservoir after aspiration. The filled liquid automatically fills the channel and replaces the contained medium.
[4] Cell seeded assays can be stored in incubator or 6-well plate with various experimental conditions.
[5] In 4 days of culture, hMVECs form intact monolayer in channel and on Type-1 collagen wall. Actin filaments and nuclei were then stained withrhodamine-phalloidin (yellow, Sigma-Aldrich) and 4’,6-diamidino-2-phenylindole (DAPI, blue, Sigma-Aldrich), respectively. Scale bar indicates 50 microns. Endothelialcell monolayer was imaged by confocal microscopeand its diffusion coefficient was measured by fluorescent dextran diffusion.


