2.1 Atoms and Atomic Bonds
Learning Outcomes
Upon completion of this section, you should be able to
1. Distinguish among the types, locations, and charges of subatomic particles.
2. Relate how the arrangement of electrons determines an element's reactivity.
3. Explain how isotopes are useful in the study of biology.
4. Contrast ionic and covalent bonds.
5. Identify the reactants and products in a chemical equation.
As you are studying right now, everything around you, including yourdesk and computer, is made of matter. Matter may be defined as anything thattakes up space and has mass. Matter can exist as a solid, liquid, gas, orplasma. Living organisms, such as ourselves, and nonliving things, such as theair we breathe, are all made of matter.
All matter is composed of elements. An element is a substance thatcannot be broken down into other substances by ordinary chemical means. There areonly 92 naturally occurring elements, and each of these differs from the othersin its chemical or physical properties, such as density, solubility, melting point,and reactivity.
While all of the elements are present on Earth, the proportion ofeach element differs between living organisms and nonliving things. Four elements—carbon, hydrogen, nitrogen, and oxygen—make up about 96% of the body weight of most organisms, from simple, one-celled life-forms to complex,multicellular plants and animals. Other elements, such as phosphorus, calcium,and sulfur, may also be found in abundance in living organisms. A number ofelements, including minerals such as zinc and chromium, are found at very low,or trace, levels. Regardless of their abundance and function in livingorganisms, the basic building blocks of each element share some commoncharacteristics.
Atomic Structure
The atomic theory states that elements consist of tiny particles called atoms. Because each element consists of only one kind of atom, the same name isgiven to an element and its atoms. This name is represented by one or twoletters, called the atomic symbol. For example, the symbol H stands for an atom of hydrogen, and the symbolNa (for natrium in Latin) stands for an atom of sodium.
If we could look inside a single atom, we would see that it is made mostlyof three types of subatomic particles: neutrons, which have no electrical charge; protons, which have a positive charge; and electrons, which have a negative charge. Protons andneutrons are located within the center of an atom, which is called the nucleus, while electrons move about the nucleus.
Since atoms are a form of matter, you might expect each atom to have a certainmass. In effect, the mass number of an atom is just about equal to the sum of its protons and neutrons.Protons and neutrons are assigned one mass unit each. Electrons, being matter,have mass, but they are so small that their mass is assumed to be zero in mostcalculations. The term massis used, rather than weight,because mass is constant but weight is associated with gravity and thusvaries depending on an object's location in the universe.
All atoms of an element have the same number of protons. This is called theatom’s atomic number. The number of protons makes an atom unique and may be used to identifywhich element the atom belongs to. As we will see, the number of neutrons mayvary between atoms of an element. The average of the mass numbers for theseatoms is called the atomic mass.
The atomic number tells you the number of positively charged protons. Ifthe atom is electrically neutral, then the atomic number also indicates the numberof negatively charged electrons. To determine the usual number of neutrons,subtract the number ofprotons from the atomic mass and take the closest whole number.

Isotopes
Isotopes are atoms of the same element that differ in the number of neutrons.
In other words, isotopes have the same number of protons, but they havedifferent mass numbers. In some cases, a nucleus with excess neutrons isunstable and may decay and emit radiation. Such an isotope is said to beradioactive. However, not all isotopes are radioactive. The radiation given offby radioactive isotopes can be detected in various ways. Most people arefamiliar with the use of a Geiger counter to detect radiation. However, othermethods to detect radiation exist that are useful in medicine and science. For example, radioactive isotopes can be used as tracers to detect molecularchanges or to destroy abnormal or infectious cells. Since both radioactiveisotopes and stable isotopes contain the same number of electrons and protons,they essentially behave the same in chemical reactions. Therefore, a researchercan use a small amount of radioactive isotope as a tracer to detect how a groupof cells or an organ is processing a certain element or molecule. For example,by giving a person a small amount of radioactive iodine (iodine-131), it ispossible to determine whether the thyroid gland is functioning properly.Another example is a procedure called positron- emission tomography (PET),which utilizes tracers to determine the comparative activity of tissues. Aradioactively labeled glucose tracer that emits a positron (a subatomicparticle that is the opposite of an electron) is injected into the body.Positrons emit small amounts of radiation, which may be detected by sensors andanalyzed by a computer. The result is a color image that shows which tissuestook up glucose and are thus metabolically active. A number of conditions, suchas tumors, Alzheimer disease, epilepsy, or a stroke, may be detected using PETscans.
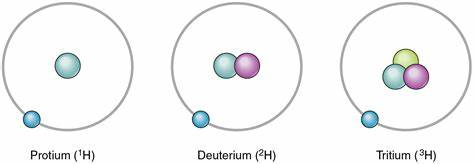
Figure 1 Isotopes of Hydrogen
Protium, designated 1H, has one proton andno neutrons. It is by far the most abundant isotope of hydrogen in nature.Deuterium, designated 2H, has one proton and one neutron. Tritium, designated3H, has two neutrons.
Types of Chemical Bonds
A group of atoms bonded together is called a molecule.When a molecule contains atoms of more than one element, it can be calleda compound. Compounds and molecules contain two primary types of chemical bonds: ionicand covalent. The type of bond that forms depends on whether two bonded atoms shareelectrons or whether one has given electrons to the other. For example, in hydrogengas (H2), the two hydrogen atoms are sharing electrons in order to fill thevalence shells of both atoms. When sodium chloride (NaCl) forms, however, thesodium atom (Na) gives an electron to the chlorine (Cl) atom, and in that wayeach atom has eight electrons in the outer shell.
Ionic Bonding
An ionicbond forms when two atoms are held together by the attraction between oppositecharges. The reaction between sodium and chlorine atoms is an example of how anionic bond is formed. Consider that sodium (Na), with only one electron in itsthird shell, usually gives up an electron (Fig. 2.7a). Once it does so, the second shell, with eight electrons, becomes itsouter shell. Chlorine (Cl), on the other hand, tends to take on an electron,because its outer shell has seven electrons. If chlorine gets one moreelectron, it has a completed outer shell. So, when a sodium atom and a chlorineatom react, an electron is transferred from sodium to chlorine. Now both atomshave eight electrons in their outer shells. This electron transfer causes theseatoms to become ions, or charged atoms. Thesodium ion has one more proton than it has electrons; therefore, it has a netcharge of +1 (symbolized by Na+). The chloride ion has one more electron thanit has protons; therefore, it has a net charge of –1 (symbolized by Cl–). Negatively charged ions often have names that end in “ide,” and thus Cl– is called a chloride ion. In the periodic table (see Fig. 2.3), atoms ingroups 1 and 2 and groups 6 and 7 become ions when they react with other atoms.Atoms in groups 2 and 6 always transfer two electrons. For example, calcium becomesCa2+, while oxygen becomes O2–.


